- Helix mullani Bland & J.G. Cooper 1861: 363, 448, pl. 4 figs 16, 17.
- Polygyra (Triodopsis) mullani var. olneyae Pilsbry 1891: 47.
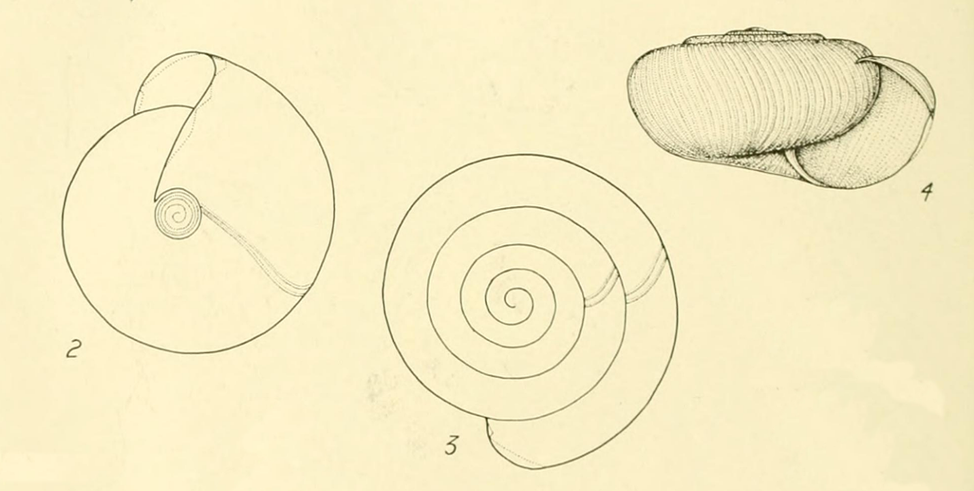
Identification. Shell depressed. Spire low-convex, low. Periphery rounded, medial. Whorls c. 5–6, convex. Suture well impressed. Protoconch with microscopic striae coarsely broken into granules. Teleoconch with incremental wrinkles/striae and weaker spiral striae, especially on base. Periostracum always without hairs (in adults). Aperture subovate-lunate, wider than high, dentulous in adults. Parietal denticle prominent, somewhat drop-shaped, elongate, and slightly curved in basal view, rather squarish in apertural view. Umbilicus c. 1/9of shell width, partly overhung by columellar lip. Lip distinctly thickened and strongly “rolled” back. Shell opaque, shining, greyish- to yellowish-brown; lip and parietal denticle white. Shell toc. 17.1 mm wide (wider than high).
Animal dark greyish brown.
Comparison. This species is quite unlike the only other Cryptomastix in BC; the shell of C. devia is more globose and less flattened.
Habitat. Moist forests; under dead wood and vegetation and in scree.
Geographic range. In BC, north to Revelstoke and west to Vernon (Pilsbry 1940) and Chase Creek near Chase (unpublished data). The historical Vernon record at first seems suspect, but with the discovery of this species along Chase Creek and in the vicinity, Vernon cannot be ruled out. However, whether habitat still exists in Vernon for this species remains to be determined.
Etymology. Cryptos (Greek), “hidden” + mastix (Greek), “flagellum”, in reference to the genital anatomy (with a very short flagellum). Mullani: after Captain John Mullan Jr (1830–1909), US Army, who built the road over the Bitter Root and Rocky Mountains in 1853–1854 (Pilsbry 1940; Wikipedia).
Remarks. The genus Cryptomastix, and certainly the group that containsC. mullani and similar species, appears in need of taxonomic revision. Following the classification of Pilsbry (1940), this species has several named subspecies. Few of these taxa are well known, and generally most have been largely ignored, relegated to obscurity due to their infraspecific rank. It is likely that at least some of these taxa are valid biological species, as already found in the genus Vespericola (e.g. Roth and Miller 1993, 1995, 2000), and all purported subspecies warrant further study. Burke (2013) provided descriptions and excellent photographs of most of these, but he did not offer any taxonomic innovation.
Specimens from Vernon, BC were classified by Pilsbry (1940) as the subspecies olneyae, which had originally proposed by him as a variety (type locality: Spokane, Washington). For now, BC records are all referred to Cryptomastix mullani s.s. For now, British Columbia populations are all referred to C. mullani (sensu lato).


References
- Bland T, Cooper JG (1861) Notice of land and fresh-water shells, collected by Dr. J.G. Cooper in the Rocky Mountains, etc., in 1860. Annals of the Lyceum of Natural History of New York 7: 362–370. https://doi.org/10.1111/j.1749-6632.1862.tb00165.x
- Burke TE (2013) Land snails and slugs of the Pacific Northwest. Oregon State University Press, Corvallis, Oregon, USA, 344 pp.
- Pilsbry HA (1940) Land Mollusca of North America (north of Mexico), Volume I, Part 2. The Academy of Natural Sciences of Philadelphia, Monographs 3: i-viii + 1-994 + i-ix.
- Pilsbry HA (1891) Polygyra (Triodopsis) mullani var. olneyae. The Nautilus 5: 47. https://www.biodiversitylibrary.org/page/42788790
- Roth B, Miller WB (1993) Polygyrid land snails, Vespericola (Gastropoda: Pulmonata), 1. Species and populations formerly referred to Vespericola columbianus (Lea) in California. The Veliger 36: 134–144. https://www.biodiversitylibrary.org/page/42465744
- Roth B, Miller WB (1995) Polygyrid land snails, Vespericola (Gastropoda: Pulmonata), 2. Taxonomic status of Vespericola megasoma (Pilsbry) and V. karokorum Talmadge. The Veliger 38: 133–144. https://www.biodiversitylibrary.org/page/42466795
- Roth B, Miller WB (2000) Polygyrid land snails, Vespericola (Gastropoda: Pulmonata), 3. Three new species from northern California. The Veliger 43: 64–71. https://www.biodiversitylibrary.org/page/42460712